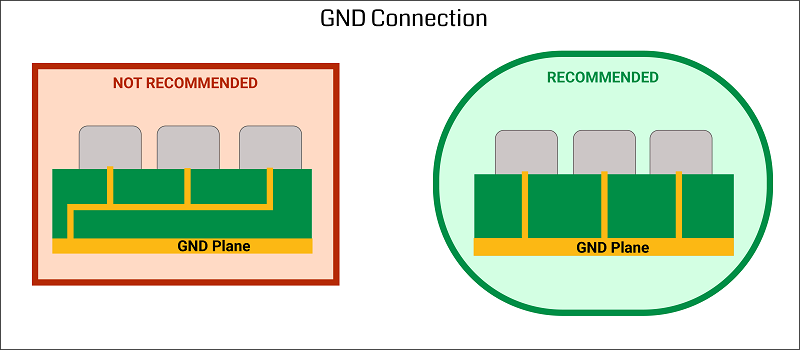
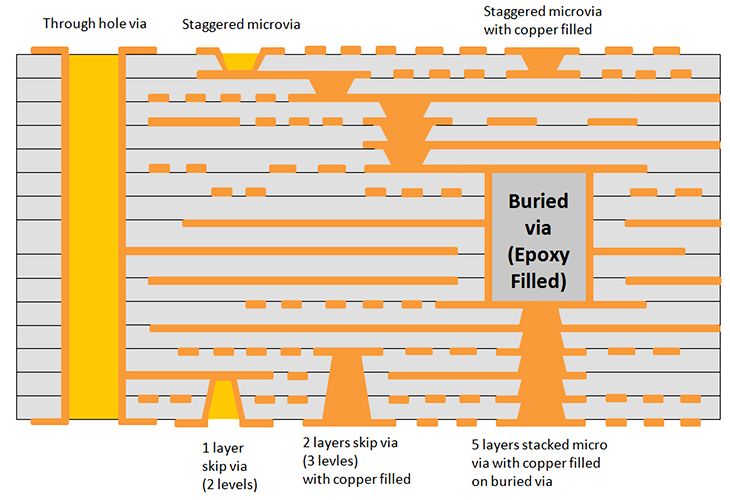
Introduction
Medical devices continue to evolve toward smaller, more efficient designs to improve patient outcomes and enable minimally invasive procedures. Rigid-flex PCBs play a pivotal role in this miniaturization trend by integrating rigid sections for mounting components with flexible sections for compact routing and folding. These hybrid boards allow engineers to pack high-density electronics into limited spaces without sacrificing reliability. In applications ranging from wearables to implants, rigid-flex PCB medical devices reduce overall device volume while maintaining structural integrity under physiological stresses. This combination addresses key challenges in medical electronics, where space constraints meet demanding performance requirements. As devices shrink, the demand for such advanced PCB solutions grows steadily.
What Are Rigid-Flex PCBs and Why Do They Matter in Medical Applications?
Rigid-flex PCBs consist of rigid substrates, typically FR4, bonded to flexible substrates like polyimide with adhesives or laminates. The design features multiple layers where rigid areas support surface-mount components and connectors, while flex tails enable bending and folding to fit irregular shapes. This structure eliminates the need for separate wiring harnesses or connectors, streamlining assembly and reducing failure points. In medical device miniaturization, rigid-flex PCB medical devices excel because they optimize space utilization in confined anatomies, such as inside the body or within handheld tools. Factory processes emphasize precise layer alignment and controlled flex thickness to ensure consistent performance. Their relevance stems from the ability to withstand repeated flexing without delamination, critical for dynamic medical environments.
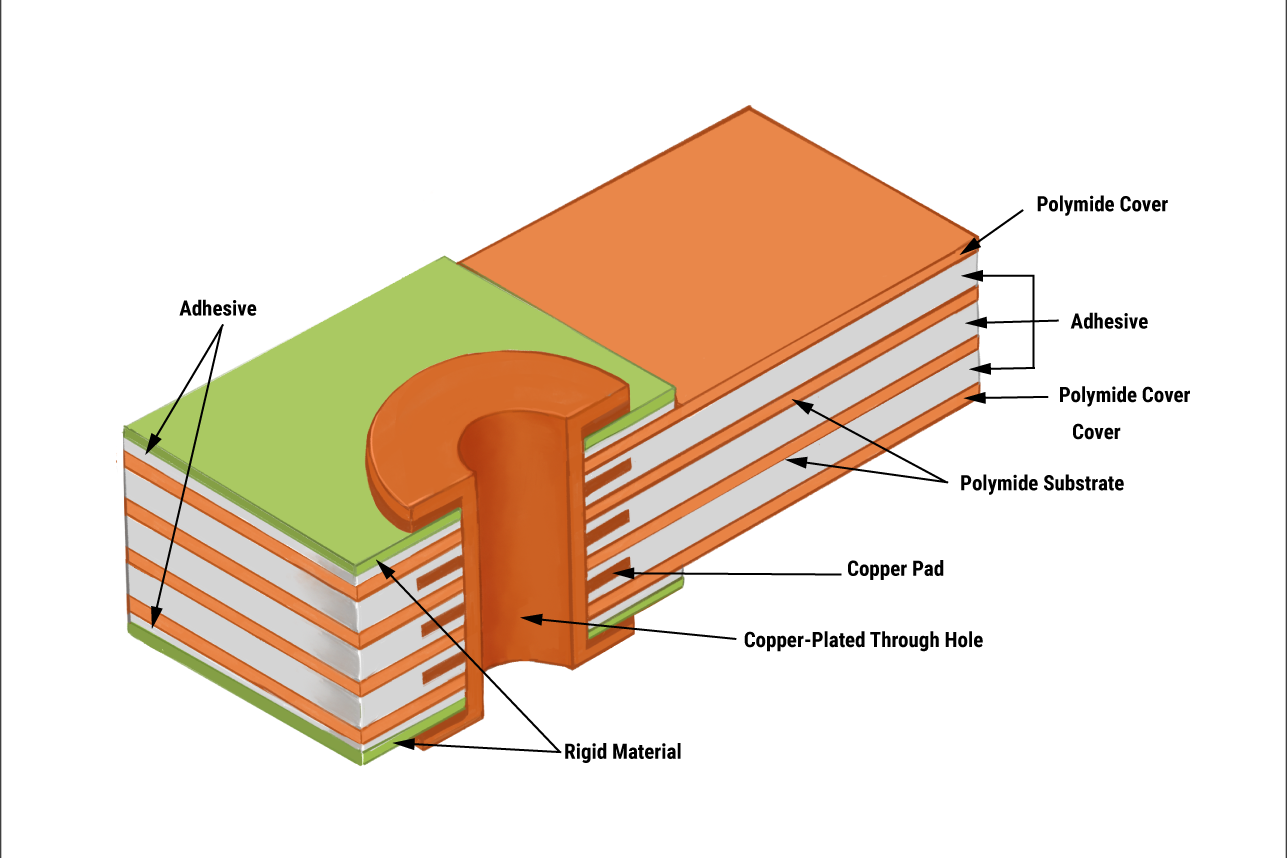
Engineers value rigid-flex boards for their role in reducing system weight and volume, which directly impacts device portability and implantation feasibility. Traditional rigid PCBs limit design freedom in curved or folded configurations, often requiring bulky interconnects that increase size. Rigid-flex solutions mitigate these issues by allowing three-dimensional layouts, making them indispensable for next-generation medical hardware. Manufacturing insights highlight the importance of material selection for long-term durability in humid, bodily fluid-exposed conditions.
Technical Principles Enabling Miniaturization
The core mechanism of rigid-flex PCBs lies in their layered construction, where rigid multilayers interlock with flexible circuits via sequential lamination. Flex areas use thinner dielectrics and copper traces optimized for bending, typically with minimum bend radii dictated by design rules. This enables folding the board to achieve up to 70% volume reduction in some layouts, though exact figures depend on specific geometries. Signal integrity remains preserved through controlled impedance in flex sections and shielding layers to counter electromagnetic interference common in medical sensors. Thermal management integrates vias and copper planes across rigid-flex transitions to dissipate heat from densely packed ICs.
Biocompatibility represents another technical pillar, as materials must resist degradation and not elicit adverse tissue reactions. Polyimide films and coverlays undergo rigorous testing to meet biological evaluation criteria, ensuring safe integration in patient-contact applications. Adhesives and soldermasks contribute to the overall inertness required for prolonged exposure. These principles align with established qualification specifications that govern flex cycle endurance and environmental resistance.

IPC-2223 provides sectional design guidelines for flexible and rigid-flex boards, emphasizing trace routing away from high-stress bend zones and proper stiffener placement. Compliance ensures the board maintains electrical continuity after thousands of flex cycles. In medical contexts, these mechanisms support higher component densities, with fine-pitch BGAs on rigid islands connected via reliable flex traces.
Applications of Rigid-Flex PCBs in Key Medical Domains
In implantable devices, rigid-flex PCB implantable devices form the backbone of pacemakers, neurostimulators, and drug delivery pumps. The rigid section houses the microcontroller and battery interface, while flex tails route to electrodes, allowing the assembly to conform to the implant's curved housing. This folding capability minimizes incision size and tissue disruption during surgery. Hermetic sealing further protects the PCB from biofluids, with materials selected for low moisture absorption.
Rigid-flex PCB medical imaging benefits portable ultrasound probes and endoscopes, where the board snakes through flexible shafts. Imaging transducers mount on rigid platforms, interconnected by flex circuits that endure articulation during procedures. This design shrinks probe diameters, enhancing maneuverability in vascular or gastrointestinal applications. Resolution improves as miniaturization allows closer sensor placement without mechanical bulk.
For sensors, rigid-flex PCB medical sensors drive wearables like continuous glucose monitors and ECG patches. Flex sections adhere to skin contours, with rigid areas for ASICs and batteries. This hybrid form factor supports wireless transmission over extended wear periods. Vital sign accuracy relies on stable traces that flex with body movement, avoiding microcracks.
Rigid-flex PCB biocompatibility ensures these applications remain safe, with coverlayers preventing ion migration into tissues. Factory-driven validation includes accelerated aging to simulate years of implantation.
Design and Manufacturing Best Practices
Engineers should start with stackup planning, defining rigid core count and flex layer thickness early to control overall bend radius. Transition zones require coverlay cutouts and anchor points to prevent peeling during lamination. Component placement favors rigid areas for thermal cycling stability, with vias staggered in flex regions per IPC guidelines. Simulation tools verify strain distribution, guiding iterations before prototyping.
Manufacturing demands sequential buildup to align layers precisely, followed by plasma cleaning for adhesion. Solder mask application on flex tails uses photoimageable materials for fine features. Qualification testing per IPC-6013E verifies performance under thermal shock, vibration, and flex endurance, essential for class 3 medical boards. Coverlay lamination pressure must stay within specs to avoid voids.
Related Reading: Medical PCB Solutions: From Design and Manufacturing to Assembly
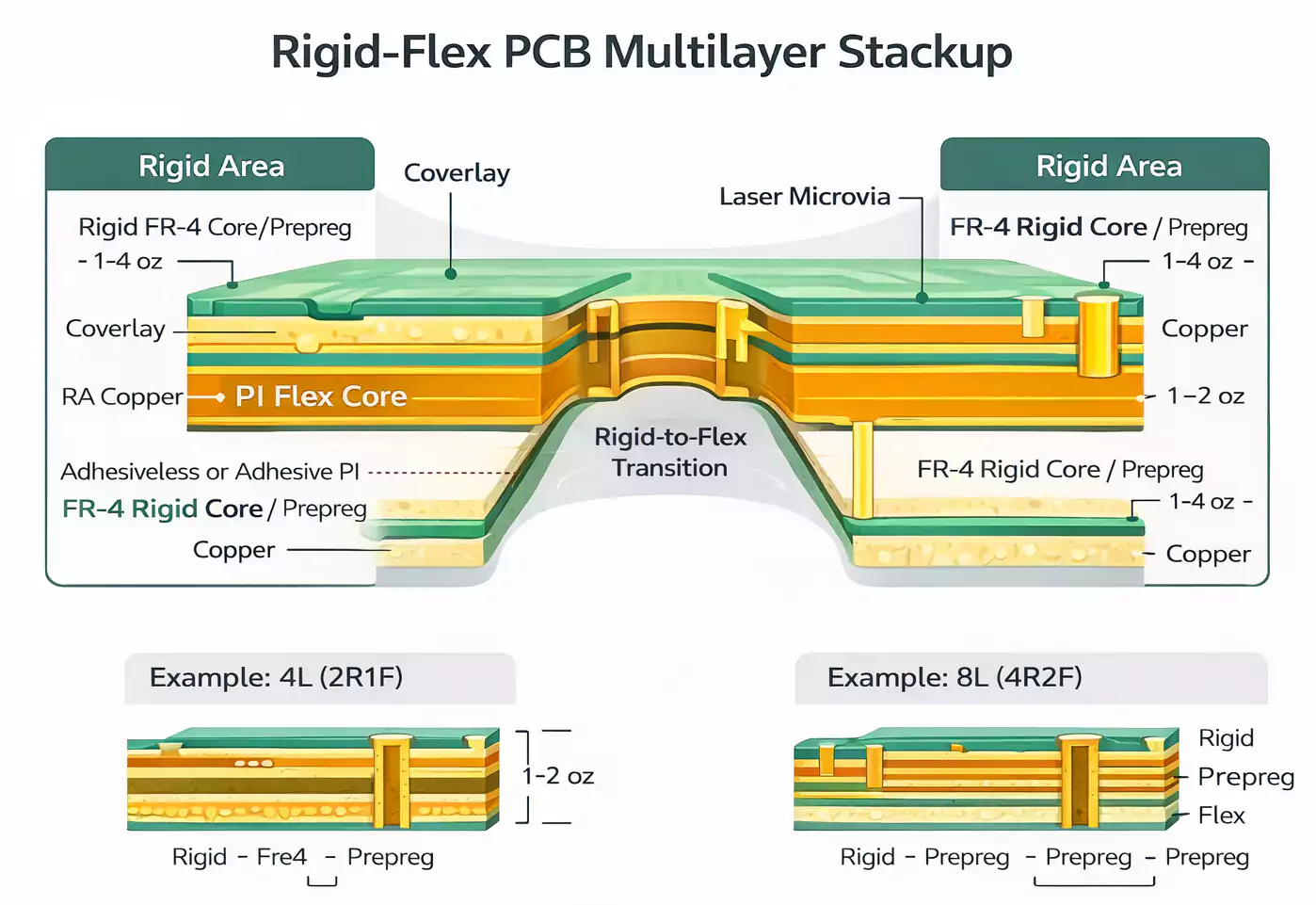
Biocompatibility protocols involve extracting materials for cytotoxicity assays, adhering to ISO 10993 frameworks. Traceability from raw polyimide to finished board supports regulatory audits. These practices minimize defects like delamination, ensuring field reliability.
Challenges and Mitigation Strategies
Dynamic stresses in medical devices can induce fatigue in flex regions, addressed by limiting bend cycles and using rolled annealed copper. Signal crosstalk in high-density flex traces requires ground planes and differential routing. Thermal expansion mismatch between rigid FR4 and flex polyimide demands low-CTE adhesives. Moisture ingress at edges calls for edge-seal epoxies post-assembly.
Factory insights recommend design reviews focusing on these transitions, with cross-section analysis confirming bond integrity. Iterative testing refines layouts for optimal miniaturization without compromising margins.
Related Reading: Decoding Medical PCB Certifications: A Comprehensive Guide
Conclusion
Rigid-flex PCBs drive medical device miniaturization by enabling compact, reliable electronics in challenging form factors. Their hybrid nature supports diverse applications from implants to sensors, underpinned by biocompatibility and performance standards. Engineers gain design freedom, reducing device footprints while upholding quality. As medical needs intensify, adherence to proven manufacturing practices will sustain innovation in this field.
FAQs
Q1: What makes rigid-flex PCB medical devices suitable for miniaturization?
A1: Rigid-flex PCB medical devices combine rigid sections for components with flexible interconnects, allowing folding to fit tight spaces like implants. This eliminates bulky wires, cuts volume, and maintains reliability under movement. Manufacturing to class 3 standards ensures endurance, aligning with factory precision for medical demands.
Q2: How does rigid-flex PCB biocompatibility impact implantable devices?
A2: Rigid-flex PCB biocompatibility requires materials like polyimide to pass biological tests, preventing tissue reactions in rigid-flex PCB implantable devices. Coverlays and adhesives contribute to inertness for long-term implantation. Standards guide evaluation, supporting safe integration in pacemakers and stimulators.
Q3: In what ways do rigid-flex PCBs enhance medical imaging equipment?
A3: Rigid-flex PCB medical imaging uses flex tails to navigate probe shafts in endoscopes, shrinking diameters for better access. Rigid islands mount cameras and LEDs, preserving image quality. This supports minimally invasive procedures with reliable signal paths.
Q4: Why choose rigid-flex for medical sensors?
A4: Rigid-flex PCB medical sensors conform to body shapes in wearables, with flex areas handling motion and rigid parts for processing. This boosts comfort and accuracy in monitoring vitals. Durability under flex cycles makes them ideal for continuous use.
 ALLPCB
ALLPCB