Introduction
Medical devices increasingly demand compact designs to enable minimally invasive procedures, wearable monitoring, and implantable technologies. Microvia PCBs play a pivotal role in achieving this miniaturization while upholding the high reliability essential for patient safety. These high-density interconnect (HDI) structures allow for finer routing and more components in smaller footprints, critical for applications like pacemakers and endoscopes. Engineers must balance density with factors such as signal integrity, thermal management, and long-term durability under physiological stresses. This article explores how microvia PCB medical devices meet stringent requirements through precise design, manufacturing, and compliance strategies. Factory-driven insights reveal the engineering rigor needed to ensure these boards perform flawlessly in life-critical environments.
What Are Microvia PCBs and Why Do They Matter for Medical Devices?
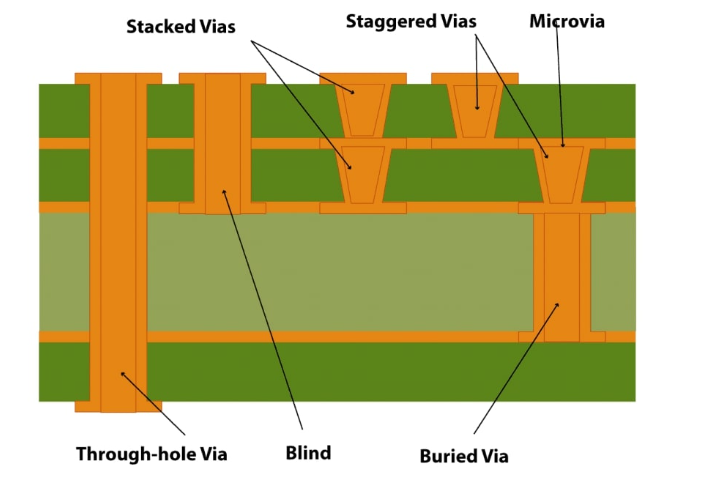
Microvia PCBs feature vias with diameters typically under 150 micrometers, enabling interlayer connections in multilayer boards without penetrating the entire stackup. Defined by industry standards, these blind or buried vias have a maximum depth of 0.25 millimeters and an aspect ratio not exceeding 1:1 to ensure reliable plating and void-free fills. In medical PCB design microvias become indispensable for shrinking device sizes while packing in advanced sensors, processors, and RF modules. For instance, portable ultrasound units or insulin pumps rely on this technology to fit complex circuitry into handheld or implantable forms. The push for miniaturization stems from patient comfort and procedural efficiency, but it introduces challenges like higher current densities and heat dissipation in confined spaces. Ultimately, microvia technology bridges the gap between performance demands and physical constraints in healthcare electronics.
Medical applications amplify the stakes, as failure rates must approach zero over extended lifecycles. Traditional through-hole vias waste board real estate and add inductance, unsuitable for high-speed signals in diagnostic imaging or telemetry. Microvias support fan-out from fine-pitch BGAs, optimizing routing efficiency by up to 50 percent in dense layers. Reliability remains paramount, with medical devices exposed to body fluids, mechanical flexing, and electromagnetic interference. Engineers prioritize these boards to comply with regulatory scrutiny while advancing device capabilities.
Technical Principles of Microvia Formation and Integration
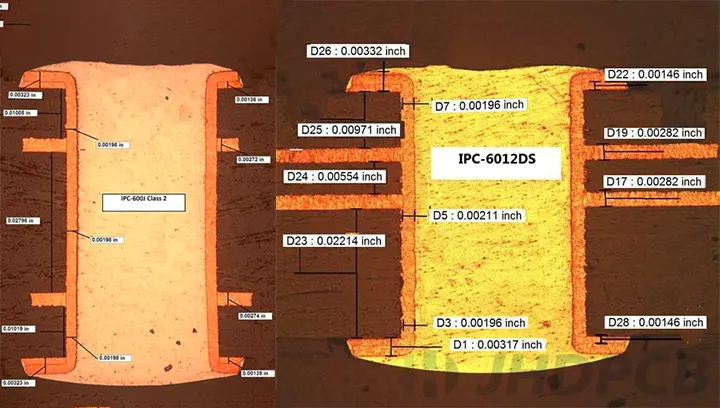
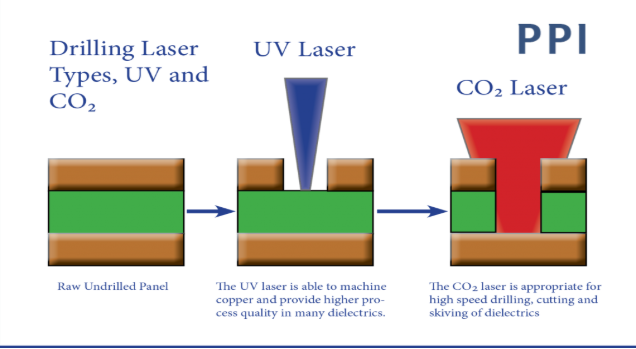
Microvias form primarily through laser ablation, where UV or CO2 lasers vaporize dielectric material to create precise holes in resin-coated copper layers. This process suits thin cores and build-up dielectrics, avoiding mechanical drill smear that plagues larger vias. Post-drilling, desmear cleans residues, followed by electroless copper seeding and electroplating to build barrel walls. Conductive pastes serve as alternatives for stacked configurations, offering cost savings but demanding thermal cycle validation. IPC-2226 provides guidelines for HDI structures, emphasizing via placement rules and capture pad sizing to prevent cracking under stress.
Stacking strategies define microvia PCB complexity: staggered vias alternate between layers for simpler processing, while stacked ones align for maximum density in any-layer designs. Aspect ratios below 0.8:1 enhance plating uniformity and reduce electromigration risks during operation. Material selection influences drillability; low-loss dielectrics with fine glass weave support clean vias without fiber pullout. Thermal expansion mismatches between copper and resin drive reliability concerns, necessitating controlled lamination cycles. Engineers model via stresses using finite element analysis to predict fatigue in medical environments.
Signal integrity benefits from microvias' shorter lengths, minimizing stubs and impedance discontinuities in high-frequency paths. However, via transitions require careful impedance control via trace width adjustments and ground plane proximity. Power delivery improves with filled vias reducing inductance, vital for mixed-signal medical circuits. These principles ensure microvia PCB high-reliability medical performance aligns with miniaturization goals.
Ensuring Reliability: Biocompatibility, Sterilization, and Durability
Microvia PCB biocompatibility hinges on substrate materials passing cytotoxicity and sensitization tests per ISO 10993 standards. While vias reside internally, encapsulants and surface finishes must prevent ion leaching into bodily tissues for implants. Flame-retardant resins and pure copper plating minimize extractables, supporting long-term implantation without inflammation. Engineers verify compliance through accelerated aging simulations mimicking physiological conditions. Factory processes incorporate cleanroom handling to avoid contamination that could compromise biocompatibility.
Sterilization poses unique challenges, as gamma radiation, ethylene oxide, or autoclaving can degrade adhesives or induce microcracks in microvias. Radiation-hardened dielectrics resist chain scission, while robust plating withstands EtO residuals. Pre-sterilization bake-outs remove moisture, preventing popcorn effects during subsequent reflows. Microvia PCB sterilization compatibility demands qualification via cycle testing, ensuring no delamination or via opens post-process. These measures safeguard device functionality throughout shelf life and usage.
High-reliability demands extend to environmental stresses: thermal cycling from -40°C to 125°C, humidity exposure, and vibration simulate implantation rigors. IPC-6012 outlines qualification for rigid boards, mandating electrical continuity and microscopy inspections on microvia integrity. Stacked vias prove vulnerable to thermo-mechanical fatigue, mitigated by via-in-pad designs and low-CTE cores. Reliability modeling predicts mean-time-to-failure, guiding stackup choices. In medical contexts, ISO 13485 enforces traceability from raw materials to final assembly, embedding quality at every step.
Related Reading: Microvia Testing: Ensuring Reliability in HDI PCBs

Best Practices for Medical PCB Design with Microvias
Start with stackup planning: limit microvia stacks to three or four to balance density and yield, using symmetric builds to curb warpage. Adhere to capture and target pad ratios per IPC-2226, typically 1.4:1 for laser vias, ensuring annular ring margins against misalignment. Route high-speed traces away from via farms to preserve integrity, incorporating stitching vias for return paths. Simulate crosstalk and PDN impedance early, iterating for compliance.
Material vetting focuses on Tg above 170°C, low Z-axis CTE under 40 ppm/°C, and laser compatibility. Sequential lamination builds outer layers post-inner via fill, allowing precise registration. Post-plating planarization via etch-back or filling preserves flatness for fine-line imaging. Factory insights stress via chain testing during fab, catching opens before assembly.
Related Reading: PCB Microvia Material Selection: Impact on Performance and Reliability
Assembly tolerances tighten for Class 3: microvia sites accept 0.3 mm pitch components, demanding precise stencil apertures. Underfill or molding protects against flex in wearables. Validation includes flying probe for nets and X-ray for BGA voids. These practices yield microvia PCB medical devices ready for regulatory submission.
Challenges and Mitigation Strategies
Warpage from CTE mismatches plagues thin HDI panels, exacerbated by microvia asymmetry. Controlled lamination with carrier plates and fiducials maintains tolerances under 50 micrometers. CAF risks in humid medical storage require ion-getter resins and tight spacing rules. Reliability screening via MSL preconditioning per JEDEC standards flags weak structures.
Yield drops with stacking complexity, offset by process windows from DOE. Cost escalates with laser time, balanced by panel utilization optimization. Engineers address these through DFM reviews, ensuring manufacturability aligns with medical rigor.
Conclusion
Microvia PCBs empower medical devices with unmatched miniaturization and density, but only through disciplined design and process control. Adhering to standards like IPC-2226 and ISO 13485 guarantees biocompatibility, sterilization resilience, and high reliability. Electric engineers benefit from structured approaches that prioritize simulation, material science, and qualification testing. As healthcare evolves toward smaller, smarter implants, microvia technology remains central to innovation. Selecting partners versed in these nuances ensures devices meet the highest safety benchmarks.
FAQs
Q1: What role do microvias play in medical PCB design for miniaturization?
A1: Microvias enable high-density routing in HDI boards, allowing finer pitch components and shorter interconnects essential for compact medical devices like wearables and endoscopes. Design per IPC-2226 limits aspect ratios to 1:1, supporting stacked configurations without reliability loss. This reduces overall board size while maintaining signal integrity, critical for battery-powered implants. Factory processes ensure void-free fills for long-term performance.
Q2: How does microvia PCB biocompatibility impact medical applications?
A2: Biocompatibility requires materials compliant with ISO 10993, preventing tissue reactions in implantable devices. Microvia structures use inert dielectrics and plating to minimize leachables, verified through extraction testing. Encapsulation further isolates vias, ensuring safety in chronic use. Engineers select low-extractable resins to meet these demands without compromising electrical properties.
Q3: What considerations ensure microvia PCB sterilization compatibility?
A3: Sterilization methods like gamma or autoclave demand robust via plating and adhesives resistant to degradation. Pre-process moisture control prevents voids during thermal exposure. Qualification involves multiple cycles with continuity checks, aligning with ISO 13485 traceability. This upholds microvia PCB high-reliability medical standards post-sterilization.
Q4: Why prioritize IPC Class 3 for microvia PCB medical devices?
A4: IPC Class 3 specifies tighter tolerances for annular rings and plating, vital for high-reliability medical environments. It mandates rigorous inspection for microvia integrity under stress. This classification supports the zero-failure ethos in patient-contact devices, from diagnostics to therapeutics.
References
IPC-2226 - Sectional Design Standard for High Density Interconnect (HDI) Printed Boards. IPC.
IPC-6012E - Qualification and Performance Specification for Rigid Printed Boards. IPC, 2015.
ISO 13485:2016 - Medical devices - Quality management systems - Requirements for regulatory purposes. ISO, 2016.
 ALLPCB
ALLPCB