Overview
Real-time detection of biomolecules is critical for in vitro diagnostics, early disease detection, and routine health monitoring. Steroid hormones such as progesterone (P4), estradiol (E2), and testosterone are essential for growth and reproduction, but their concentrations in biological fluids are often extremely low (< nM). Clinical hormone assessment typically relies on invasive blood sampling that requires trained personnel and complex instruments. Saliva offers a noninvasive alternative, but free hormones in saliva represent only about 1% to 2% of total blood hormone levels, which limits practical clinical applications.
There is a clear need for hormone assays with ultralow detection limits, wide dynamic ranges, and simple operation to enable dynamic monitoring across several orders of magnitude. Researchers at the School of Materials Science and Engineering, Sun Yat-sen University, led by Associate Professor Zhuang Xie, have reported a three-dimensional microstructured flexible polymer electrode approach that enables low-cost, sub-femtomolar electrochemical immunosensing of sex hormones. The sensor can directly detect highly diluted saliva samples without centrifugation, facilitating at-home saliva testing and monitoring of salivary P4 and E2 across the menstrual cycle.
Sensor Design
The core sensor consists of a flexible micropyramid array coated with a conductive polymer blend of PEDOT:PSS doped with lithium bis(trifluoromethanesulfonyl)imide (LiTFSI), and gold nanoparticles (AuNPs) to support antibody immobilization. The immunosensing working electrode uses microneedle-like pyramid tips to promote diffusive enrichment of target molecules. Detection is performed by differential pulse voltammetry (DPV), where target binding causes a decrease in peak current, enabling P4 and E2 measurement across a wide range from sub-femtomolar to micromolar levels within minutes.
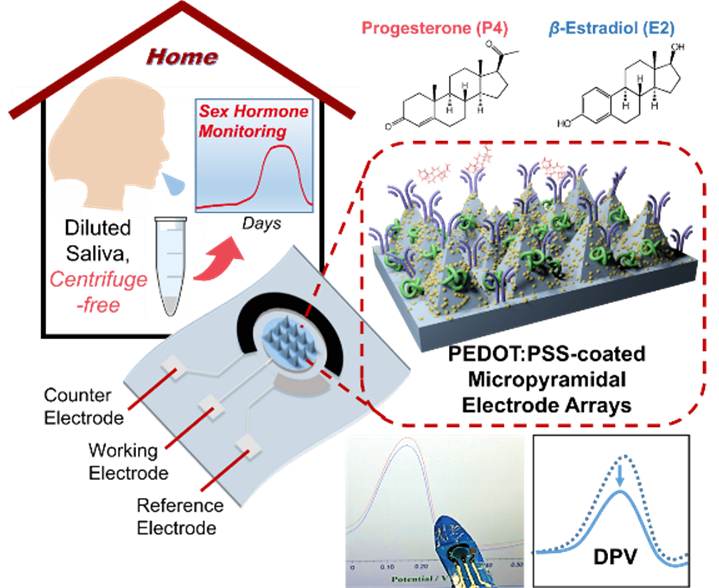
Figure 1. Flexible PEDOT:PSS-coated micropyramid array as the working electrode. The device enables electrochemical immunosensing of salivary progesterone (P4) and estradiol (E2) using DPV for sensitive steroid recognition.
Electrochemical and Simulation Characterization

Figure 2. (A) SEM image of the micropyramid array coated with PEDOT:PSS/LiTFSI. (B) Finite element simulation of the diffusion profile of Fe(CN)6 4- around the micropyramid electrode array under overpotential. (C) Three-dimensional local current density distribution for a single micropyramid electrode and the enhancement of current and area with size variation. (D-E) Comparison of cyclic voltammetry (CV) and DPV current signals between the micropyramid array and planar PEDOT:PSS/LiTFSI electrodes.
Electrode Fabrication and Analytical Performance
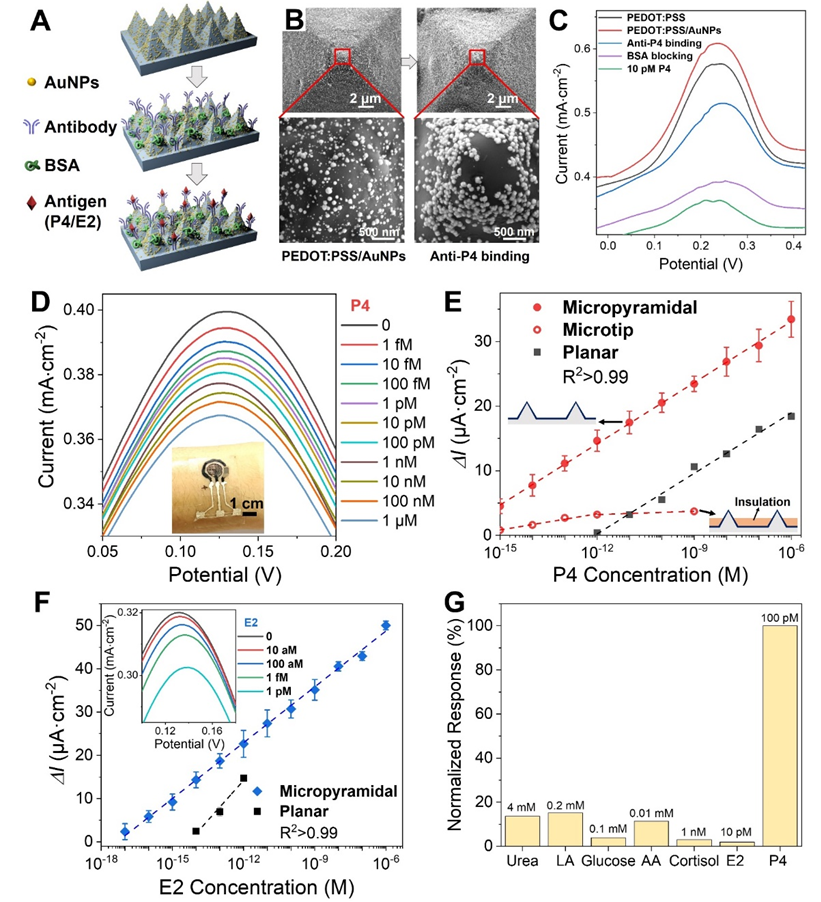
Figure 3. (A) Schematic of immunosensing electrode fabrication. (B) SEM images after electrodeposition of AuNPs (left) and after incubation with anti-P4 antibody (right) on the micropyramid PEDOT:PSS electrode. (C) DPV changes during electrode modification. (D) Typical DPV responses for P4 detection across 1 fM to 1 uM. (E) Calibration curves of DPV peak current change (Delta I) versus P4 concentration for micropyramid, microneedle-tip, and planar electrodes. (F) DPV curves and calibration for an E2 antibody-modified immunosensor. (G) Normalized DPV responses to common interferents at physiological saliva concentrations, demonstrating good selectivity of the immunosensing electrode.
Mechanical Stability and Storage
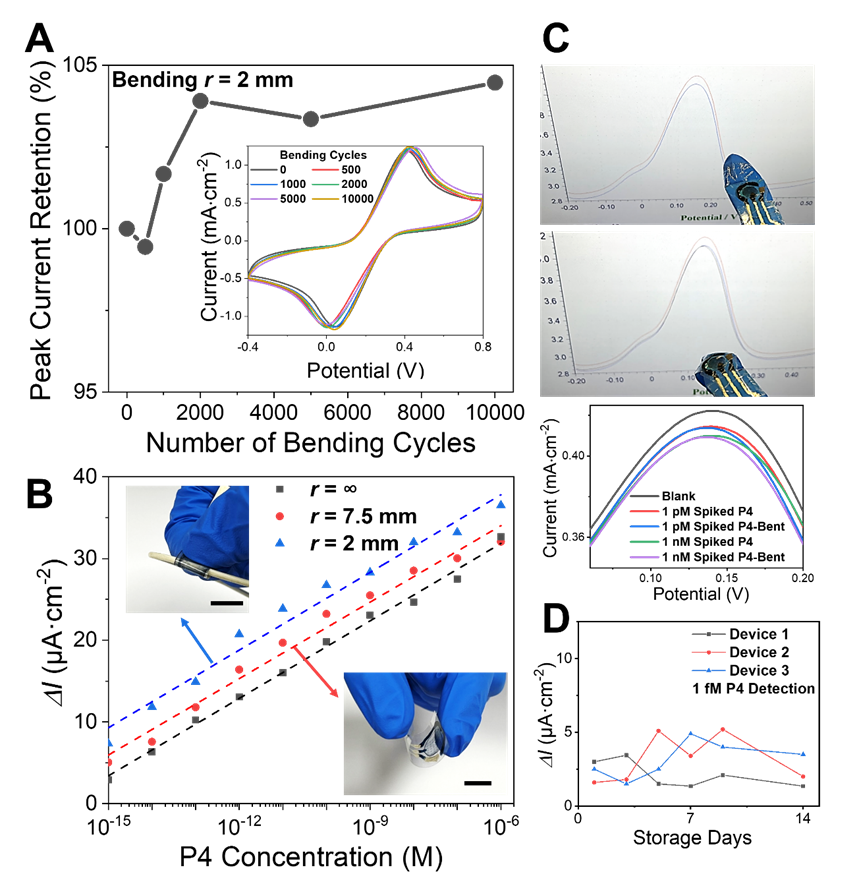
Figure 4. (A) CV peak current of the micropyramid electrode remains essentially unchanged after 10,000 repeated bending cycles at a curvature radius of 2 mm. Inset: representative CV curves during bending cycles. (B) P4 calibration curves obtained under different bending conditions. All scale bars are 1 cm. (C) The sensor mounted on a glove was used to test P4, showing negligible interference from finger bending; corresponding DPV peak current changes are negligible. (D) Devices stored at 4°C for 14 days retained the ability to detect 1 fM P4.
At-Home Saliva Testing and Monitoring
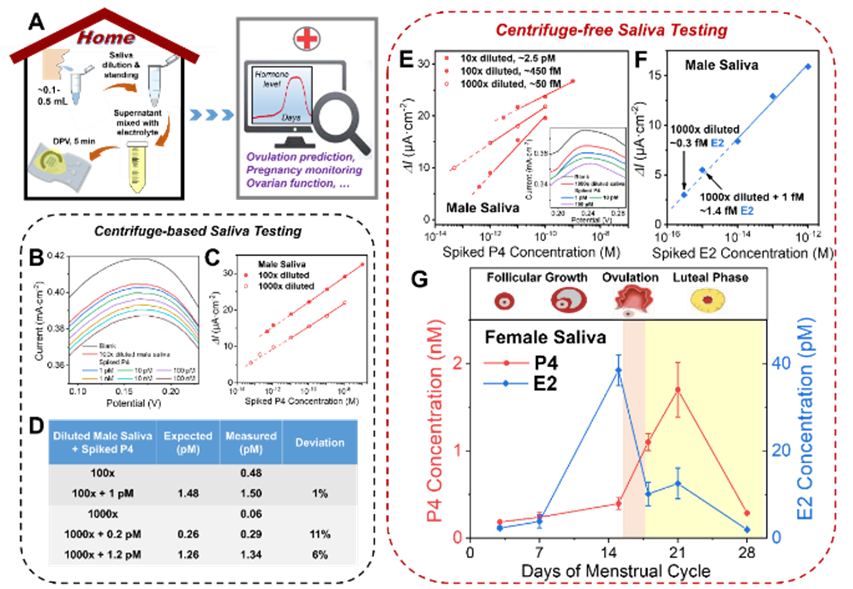
Figure 5. (A) Schematic of a no-centrifuge at-home saliva hormone testing scheme for female health monitoring. (B–D) Results for male saliva samples after centrifugation, including DPV traces for P4 spiked into 100x diluted saliva and calibration curves and detection accuracy for 100x and 1000x diluted samples. (E) No-centrifuge testing: typical current responses for a series of spiked P4 concentrations in 10x to 1000x diluted male saliva, used to determine sample background P4. Inset: DPV curve for a 1000x diluted sample. (F) Calibration curve for E2 spiked into 1000x diluted male saliva. (G) No-centrifuge saliva testing used to monitor P4 and E2 levels in 1000x diluted female saliva samples over 4 weeks. Distinct hormone level changes correspond to menstrual cycle stages and can indicate the transient ovulation window.
Conclusion
This work demonstrates a flexible electrochemical immunosensor based on PEDOT:PSS/AuNPs 3D micropyramid electrodes that achieves sub-femtomolar detection of steroid hormones. By exploiting mass-transport enhancement from the 3D microstructure, the limit of detection was reduced by approximately 1000-fold while maintaining a wide linear range from sub-fM to uM. The ultralow detection limit is well suited for measuring ultra-low concentrations of steroid hormones in highly diluted human saliva and enables noninvasive, centrifuge-free at-home monitoring of female hormone levels.
The proposed 3D micropyramidal polymer electrode platform can be further explored as a general biosensing approach. Potential applications include precise clinical diagnostics and consultation, in vivo detection of bioactive species, and tracking contaminants in food and the environment. The ultrasensitive, flexible 3D microstructured electrode platform provides a foundation for a broad range of applications.
 ALLPCB
ALLPCB