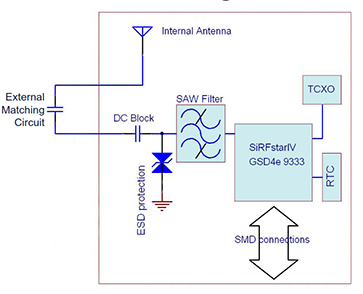
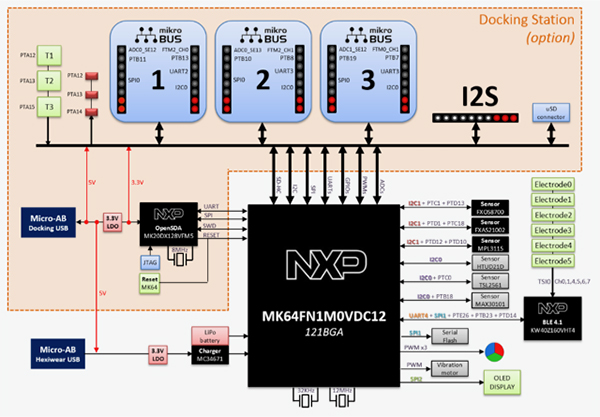
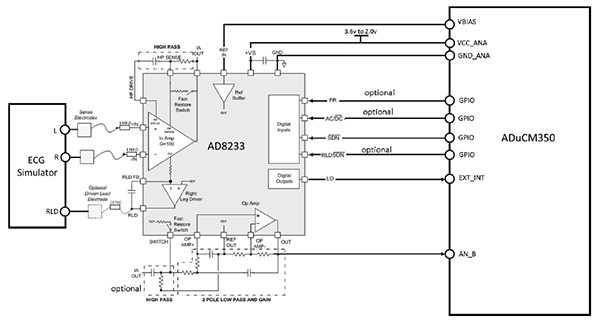
Several studies have found that concentrations of many biomarkers in sweat correlate with corresponding circulating analytes in blood. Continuous monitoring of these sweat biomarkers can enable early detection of conditions such as cystic fibrosis, diabetes, and gout by tracking chloride, glucose, uric acid, and tyrosine levels. In addition, tracking sweat loss can provide timely feedback to athletes, military personnel, and clinical caregivers to prompt rehydration and help prevent dehydration or heatstroke.
Collecting, capturing, and subsequently analyzing sweat samples from different body locations at defined time points is critical. This need has driven the development of electrochemical and colorimetric sweat sensors. Compared with electrochemical sensors, colorimetric sweat sensors do not require electronic components for data analysis and transmission, making them more compatible, lower cost, and easier to use. Colorimetric sensors are often integrated with microfluidic devices to detect multiple analytes on a single platform. By connecting microfluidic channels to a series of isolated reservoir chambers, time-series sampling and analysis of sweat can be achieved while minimizing cross-contamination or evaporation. Thus, appropriate microfluidic design makes reliable colorimetric monitoring of biomarker concentration changes in sweat possible.

Figure 1. Schematic of a skin-interfaced colorimetric microfluidic device with colorimetric detection and a microfluidic network for sweat analysis and advanced sweat control and feedback.
Timed sampling behavior of colorimetric microfluidic sweat devices
In electrochemical sweat sensors, circuitry is typically used to electronically program the microfluidic valve system for active biofluid management and to convert collected biofluids into electrical signals that can be wirelessly transmitted or displayed on a smartphone or electrochromic displays.
Colorimetric sweat sensors lack electronic components and rely on natural sweat pressure generated by perspiration or osmotic flow to drive sweat through flexible microfluidic structures for sampling. Therefore, designing valve structures within microfluidics to actively manage biofluid flow for timed sampling and analysis is essential. Common designs include capillary burst valves, hydrophobic valves, and polymeric valves. Each valve type has its own advantages and limitations and plays a distinct role in timed sampling and colorimetric readout applications. Different valve types can be combined to achieve synergistic, noninvasive, in situ sweat monitoring. For example, passive polymer valves can route sweat to desired reservoirs for analysis and block channels after triggering to prevent backflow, but they often need to be paired with other valve types (capillary burst or hydrophobic valves) for timed sampling.
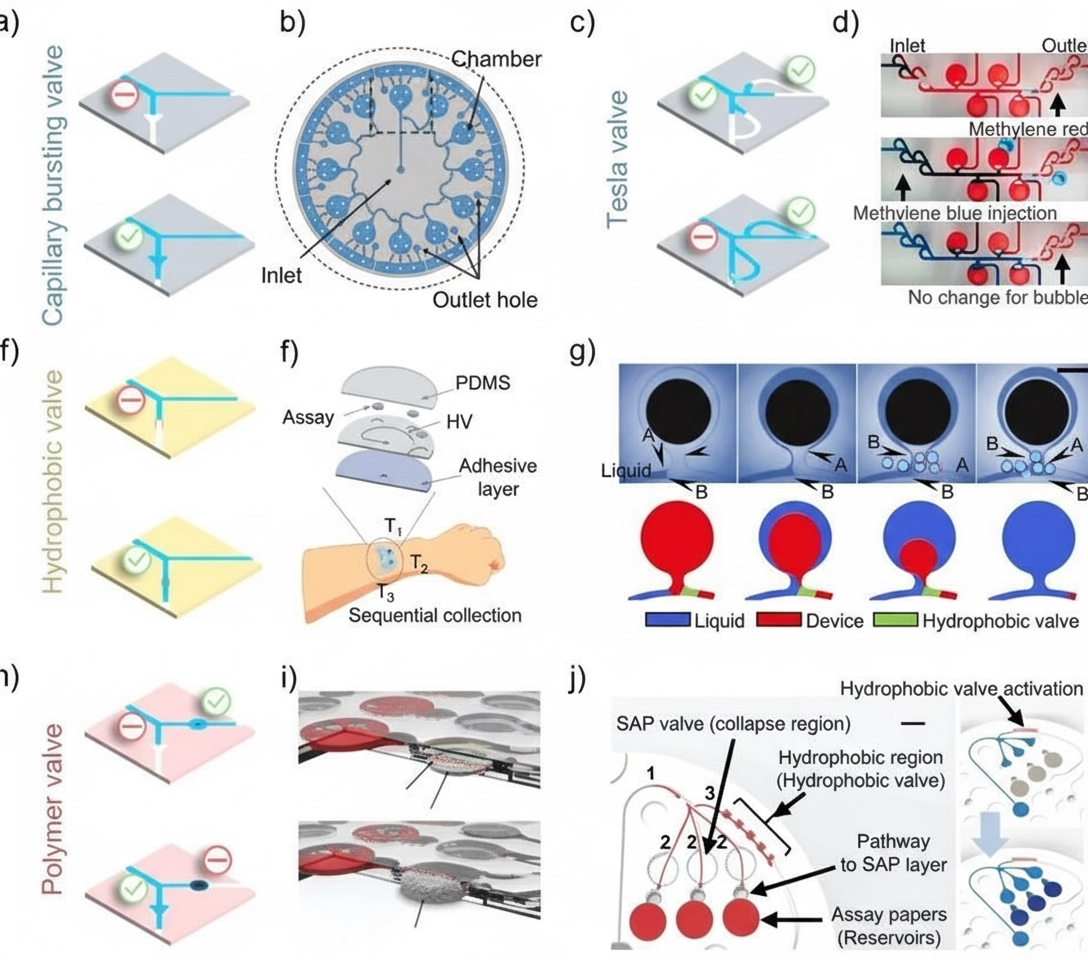
Figure 2. Skin-interfaced microfluidic device based on passive valve structures for timed sweat sampling.
Electronic-free sweat control and feedback techniques
Main challenges for colorimetric microfluidic devices include: (1) uncontrolled sweat flow and mixing; (2) soluble chemical reagents potentially backflowing from reaction chambers onto the skin; (3) uncertainty in precise analysis timing due to variable sweat rates; (4) difficulty performing multi-step colorimetric assays; (5) irreversible colorimetric reactions that prevent continuous monitoring; and (6) lack of timely self-feedback mechanisms. One potential solution is integrating the sweat analysis system with electronically programmable microfluidic valves that use individually addressable microheaters to control sweat flow through microchannels blocked by thermoresponsive hydrogels. Active valve control enables sweat analysis at user-defined times, independent of sweat flow rate and other external disturbances.
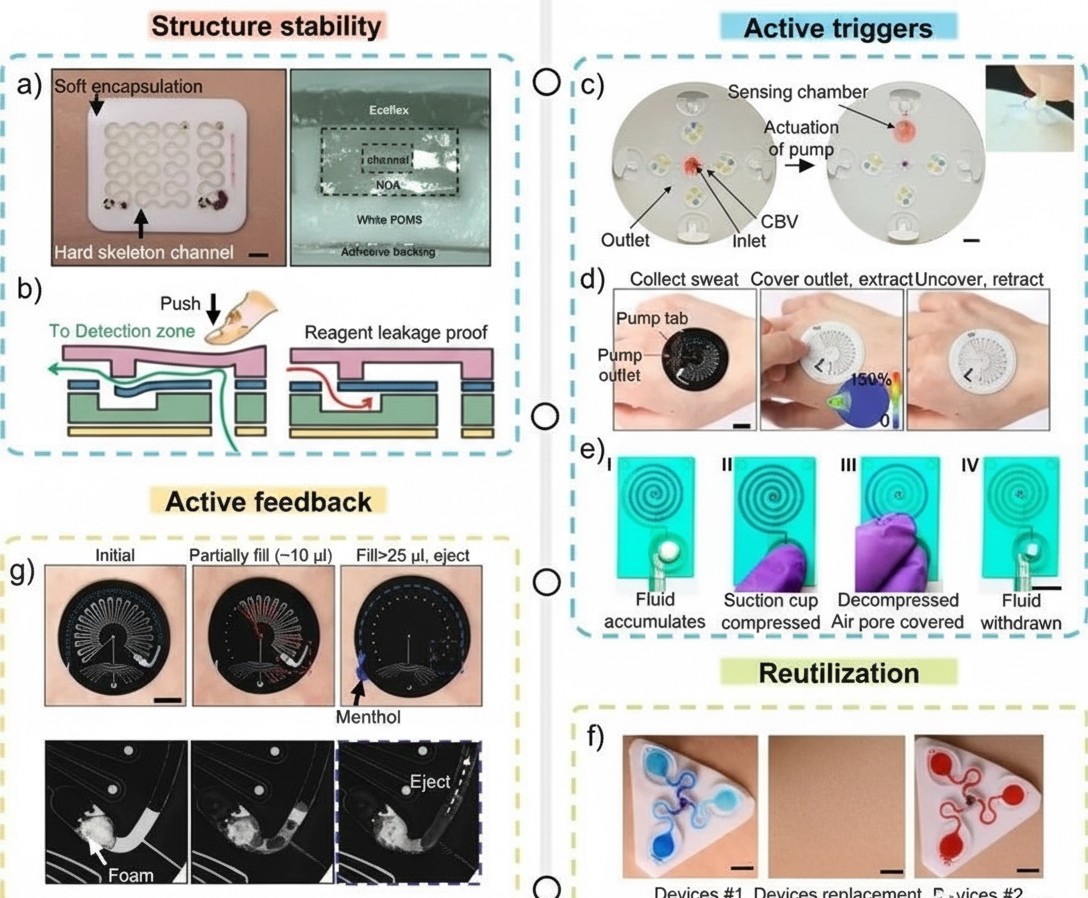
Figure 3. Electronic-free sweat control and feedback techniques in colorimetric microfluidic devices.
Overall, although research on colorimetric sweat sensors has progressed significantly, many challenges remain to achieve higher comfort, stable function under mechanical deformation, and reliable low-cost manufacturing for diverse applications. First, to reduce discomfort and iatrogenic injury, wearable devices—particularly those intended for populations with delicate skin such as infants—should be flexible and deformable. However, device deformation during use or other nonspecific external factors can affect sweat flow behavior in microfluidic devices and may unintentionally trigger feedback components. While integrated microfluidic devices with relatively rigid microchannels on flexible substrates can mitigate this issue, their fabrication is often expensive and time-consuming (for example, photolithography and deep reactive ion etching with packaging times exceeding 16 hours), and their materials and moduli are poorly matched to skin. In contrast, digital light processing (DLP) can rapidly fabricate (less than 1 hour) 3D-printed channels with micron-scale feature sizes (<100 μm) and improved optical transparency.
At the same time, the gray-scale range of DLP enables fabrication of gradient functional materials with mechanical gradients spanning up to three orders of magnitude by using different light intensities, which helps alleviate abrupt modulus transitions. Second, accurate sweat analysis using skin-interfaced wearables requires reducing contamination from skin secretions such as sebum, skin debris, and dust. For example, researchers designed a paper-based sandwich pH sensor that filters sebum mixed into sweat using common oil blotting pads. Finally, most colorimetric sweat sensors also face issues with single-use reagents and discontinuous detection. Until reversible colorimetric reagents are available, replaceable colorimetric modules remain a viable alternative.
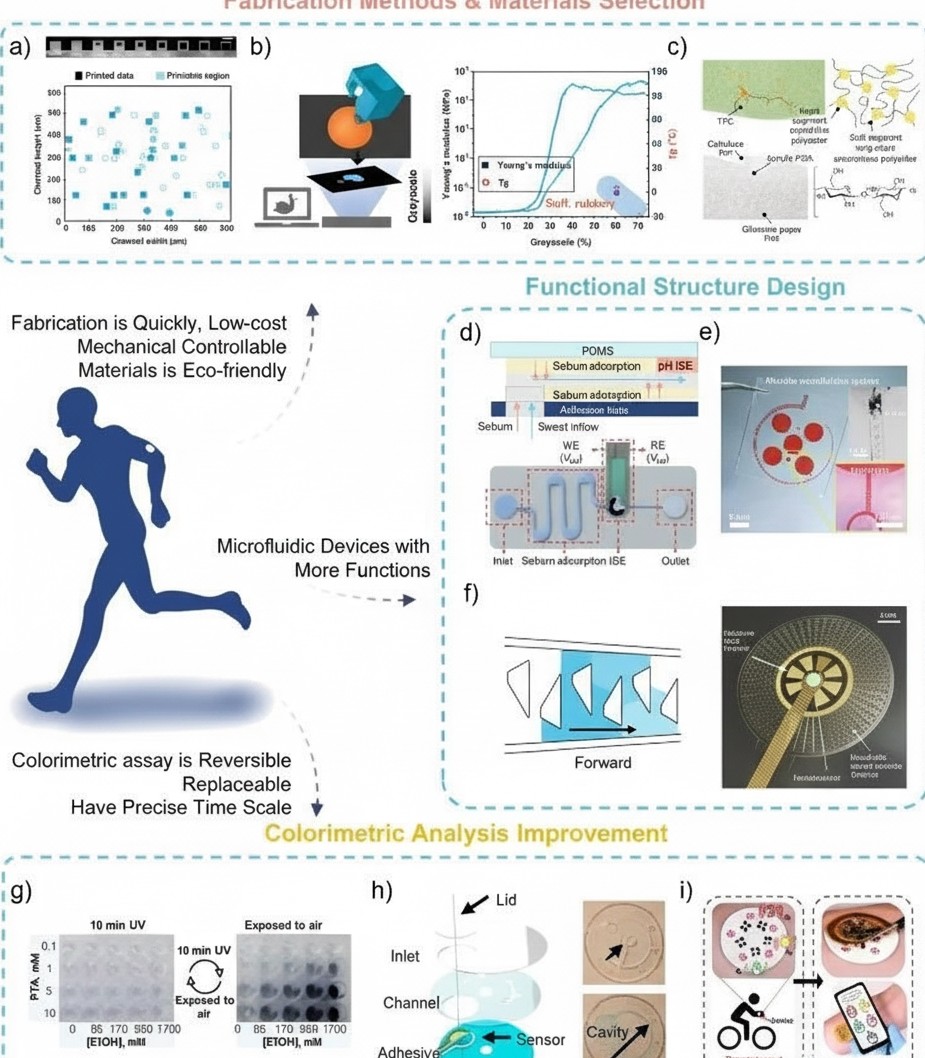
Figure 4. Future opportunities and challenges for wearable colorimetric sweat monitoring systems.
 ALLPCB
ALLPCB