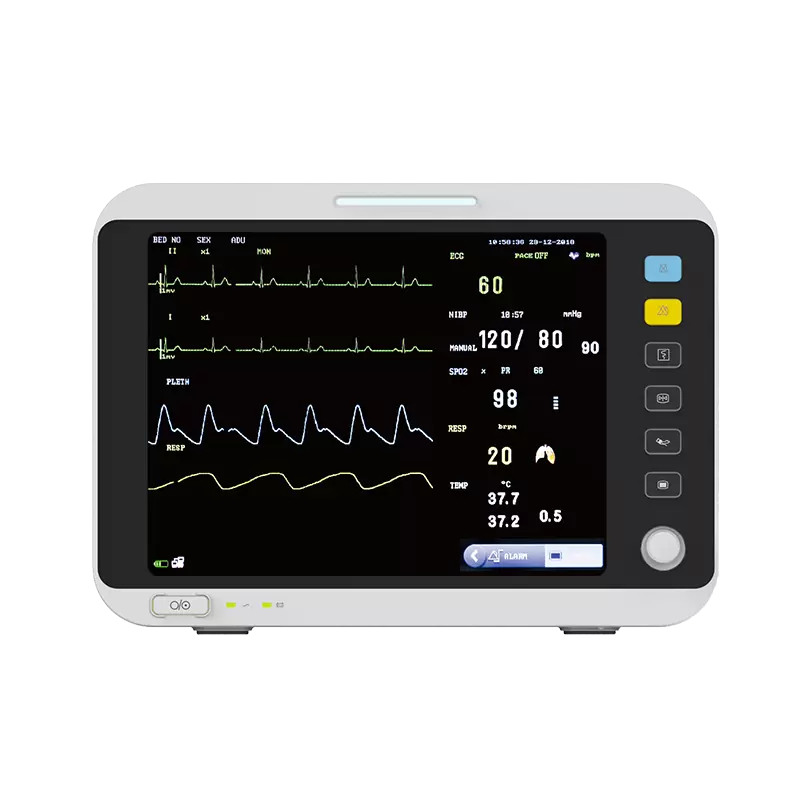
Overview
NTC thermistors can be used for temperature sensing, while RFID tags enable inventory control of medical supplies. This article summarizes Murata Manufacturing's NTC thermistors and RFID products and outlines their key characteristics for reference in related product development.
NTC Thermistors as Temperature Sensors
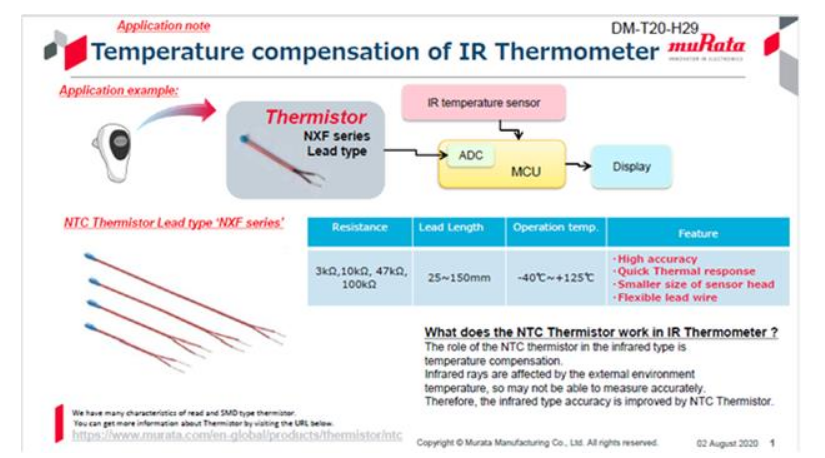
NTC (negative temperature coefficient) thermistors are resistive devices whose resistance decreases as temperature rises. They are manufactured from sintered non-oxide ceramic materials containing manganese (Mn), nickel (Ni), cobalt (Co), and other elements, with electrodes formed on the ceramic. Common package styles include leaded and SMD chip types. Resistance variation with temperature typically ranges from about 1% to 5% per °C, making NTC thermistors a common choice for temperature sensing in electronic devices.
Murata's NXF series are leaded NTC thermistors designed for high precision and good thermal response. The product range includes SMD chip and leaded variants for applications such as temperature detection, overheat detection, circuit protection, current control, and heating. The NXF series uses flexible lead wires with lengths from 25 to 150 mm, offering compact size and fast response suitable for electronic thermometers.
RFID Tags for Medical Equipment Management
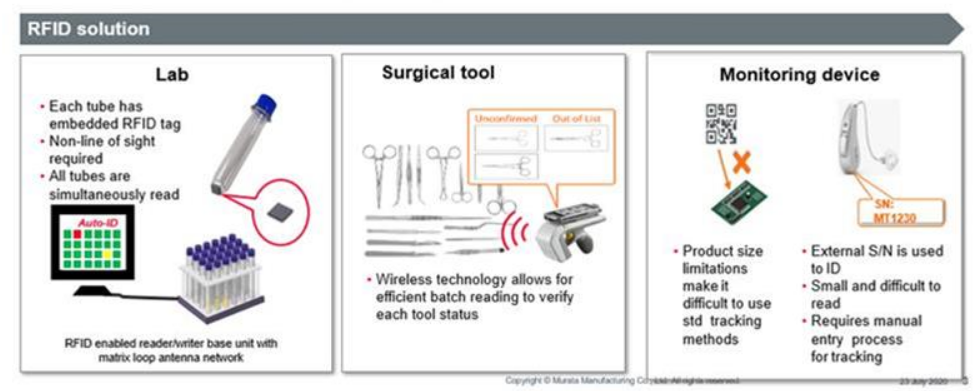
During surgical procedures, many different instruments are used, and managing these instruments is challenging. U.S. regulations required unique device identifiers (UDIs) on surgical instruments by 2020, and similar regulatory requirements are being implemented in other regions with phased timelines extending into the coming years. Manual recordkeeping is time consuming and error prone, and assembling and verifying instrument sets requires significant labor even for experienced staff.
Traditional methods such as barcodes, laser marking, or QR codes have limitations: barcode reading on surgical instruments is often difficult, laser marking can promote corrosion or contaminant adhesion, and QR code scanning is slow. RFID tags provide a more practical option: they can be attached to a wide range of medical devices, store and update device information such as usage time and lifetime, enable efficient inventory audits through batch reads, and allow reading even when instruments are contaminated, reducing manual effort.
Batch RFID reads accelerate instrument set assembly and verification. Usage frequency for each instrument can be recorded and displayed on PCs or other systems, enabling optimization of preoperative instrument preparation and potential inventory reductions based on measured usage.
Murata's LXTBKZMCMG-010 is a UHF RFID tag designed for surgical instruments and medical devices with metal surfaces. It is a compact RFID module intended for placement on metal objects and supports global use. The LXTBKZMCMG-010 uses the UHF band (865-928 MHz) and complies with ISO 18000-63 / EPCglobal Gen2 (v2). Its dimensions are 6.0 x 2.0 x 2.3 mm, it uses the Impinj Monza R6P IC, and on metal it can achieve a maximum read range of up to 150 cm (with 4 W EIRP). The module is RoHS compliant.
These RFID tags for metal applications are specified to withstand autoclave sterilization processes. Tags can be read using handheld readers or fixed readers with antennas to perform real-time reads of instruments on a workspace without manual scanning. Typical read ranges vary from about 10 cm to 1 m depending on the environment and setup.
Traceability for Small Products
Many small, customizable products such as hearing aids require individual recording of manufacturing records and device settings. Because these products are miniature, conventional methods like QR codes become impractical. RFID tags can be embedded or attached so that product information is readable from outside the device, and the data can be updated. RFID is resistant to contamination compared with surface labels that may become soiled through skin contact.
Murata's LXMSJZNCMF-198 and LXMS21NCNH-147 are UHF RFID modules intended for tracking small products such as hearing aids, smart trackers, medical cases, and laboratory items. These modules can be applied to metal and non-metal surfaces or embedded using adhesive, double-sided tape, or overmolding, and are designed for high reliability.
The LXMSJZNCMF-198 is a compact, robust module using the UHF band (865-928 MHz) and complying with ISO 18000-63 / EPCglobal Gen2 v2. Its dimensions are 1.2 x 1.2 x 0.55 mm, it uses the Impinj Monza R6 IC, and it provides a read range of approximately 10 mm. The LXMS21NCNH-147 has similar characteristics but complies with ISO 18000-63 / EPCglobal Gen2 (v1.2.0), measures 2.0 x 1.25 x 0.55 mm, and uses NXP's G2iM IC.
Small, robust RFID modules can be mounted on PCBs and overmolded so that device history tracking begins at manufacturing. Antenna design and tag integration can be supported to improve production workflow and post-sales support efficiency.
RFID for Medication Safety
Medication errors are a persistent concern in healthcare. Applying RFID tags to medications, for example to ampoules, helps prevent administration errors and enables automatic recording of doses, checks for expiration, and verification of correct medication. RFID tags can be embedded in the product rather than on external packaging, allowing identification even after a drug is removed from its outer box. Each tag can store device settings and prescription history in re-writable memory.
RFID tags can include password locking to protect stored data, and each IC typically has a unique, non-rewritable serial number for product verification. Tags can be attached by adhesive, double-sided tape, or overmolding. Non-contact reading avoids mechanical wear, and communication between readers and tags can be maintained even if surfaces are contaminated by chemicals or other substances.
Conclusion
NTC thermistors are suitable for temperature sensing in electronic thermometers and other medical applications. UHF RFID tags provide capabilities for equipment tracking, inventory management, and medication safety by improving identification accuracy, reducing human error, and enabling lifecycle traceability including return material authorization tracking. These technologies can support process efficiency and product traceability in medical settings.
 ALLPCB
ALLPCB